 *Vital notice: bioRxiv publishes preliminary scientific reports that usually are not peer-reviewed and, due to this fact, mustn’t be thought to be conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Vital notice: bioRxiv publishes preliminary scientific reports that usually are not peer-reviewed and, due to this fact, mustn’t be thought to be conclusive, guide clinical practice/health-related behavior, or treated as established information.
A team of scientists from Australia and Israel demonstrates that Candida auris, a fungus related to life-threatening and drug-resistant infections, escapes the host’s innate immune response by destroying macrophages through metabolic reprogramming. The fungus can be able to escaping antimicrobial inflammatory response.
The study is currently available on the bioRxiv* preprint server.
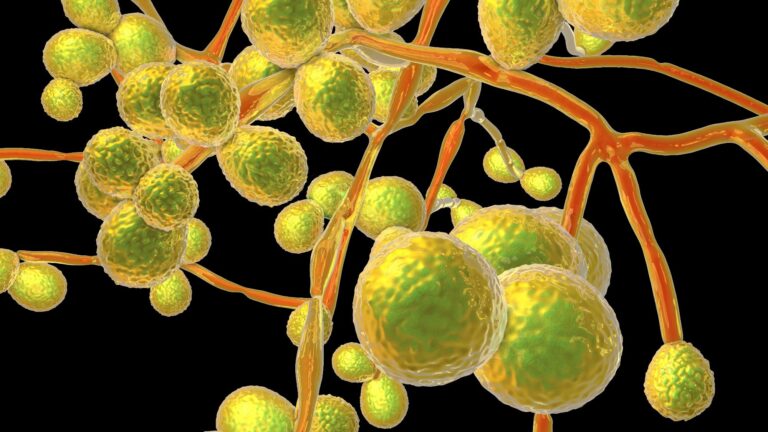
 Study: Candida auris evades innate immunity by utilizing metabolic strategies to flee and kill macrophages while avoiding antimicrobial inflammation. Image Credit: Kateryna Kon / Shutterstock
Study: Candida auris evades innate immunity by utilizing metabolic strategies to flee and kill macrophages while avoiding antimicrobial inflammation. Image Credit: Kateryna Kon / Shutterstock
Background
Candida auris is an emerging fungus liable for blood infections with a high mortality rate. The infections are multidrug-resistant and pan-drug-resistant in nature.
Some studies have suggested that fungal cell partitions might play a task in immune evasion by shielding the fungus from recognition by macrophages. Nonetheless, some studies have contradicted these findings by showing that Candida auris cell wall induces more robust pro-inflammatory responses in hosts in comparison with other common fungal pathogens. Such lack of understanding on the mode of motion of Candida auris makes it difficult to develop novel therapeutics with improved anti-fungal activity.
In the present study, scientists have investigated the mechanisms adopted by Candida auris to flee host immune responses and establish infection.
Vital observations
The study findings revealed that Candida auris escapes immune recognition and containment by macrophages and causes macrophage destruction by inducing metabolic stress. By analyzing isolates of 4 different pathogenic clades of Candida auris, the scientists observed that macrophage escape and destruction by the fungus is a conserved mechanism.
Specifically, Candida auris was found to destroy macrophages by disrupting glycolytic metabolism. Concurrently, the fungus showed the flexibility to flee recognition and elimination by the NLRP3 inflammasome. The NLRP3 inflammasome is an important component of the innate immune system that mediates the activation of an inflammatory protease (caspase-1) and induces the secretion of pro-inflammatory cytokines in response to microbial infection.
Using Candida auris-infected mice, the scientists observed that the deletion of transcriptional activator TYE7 results in the inhibition of its glycolytic metabolism, reduction of its ability to destroy macrophages, and inhibition of its ability to ascertain infection within the kidneys. These observations indicate that TYE7 serves as a significant transcriptional regulator of Candida auris metabolism and pathogenesis.
Using a live-cell imaging platform, the scientists showed that Candida auris starts escaping macrophage-mediated recognition after 8-10 hours of challenge. Furthermore, they found that Candida auris initiates macrophage destruction after 16-18 hours of challenge.
Further mechanistic evaluation revealed that Candida auris doesn’t induce macrophage lysis. As a substitute, it was observed that the glycolytic metabolic capability of the fungus supports its proliferation in macrophages, resulting in a progressive increase in fungal loads to a certain threshold to trigger the non-lytic destruction of macrophages.
Such non-lytic escape from macrophages might explain the absence of NLRP3 inflammasome activation in response to Candida auris infection. The findings revealed that due to the non-lytic mechanism, the phagosomal membrane stays intact during escape. Due to this fact, the rupture of the phagosomal membrane by invasive hyphal growth of the fungus is required to activate the NLRP3 inflammasome.
Based on these observations, the scientists suggest that the metabolic adaptations allow Candida auris to grow inside macrophages and induce non-lytic escape, which collectively contribute to its escape from antimicrobial inflammatory responses.
The scientists observed immunometabolic reprogramming of macrophages, resulting in increased glycolytic metabolism. For macrophages, it’s a conserved mechanism in response to pathogens. Nonetheless, they didn’t observe any induction in glycolysis in peripheral blood mononuclear cells.
As mentioned by the scientists, “each human and mouse macrophages undergo immunometabolic reprogramming in response to Candida auris, which renders them at risk of glucose competition by Candida auris that causes immune cell death.”
Study significance
The study demonstrates that Candida auris undergoes metabolic reprogramming to flee recognition by macrophages. This puts macrophages under metabolic stress, resulting in macrophage cell death. Nonetheless, despite macrophage destruction, no activation of the NLRP3 inflammasome has been observed, which is likely to be as a consequence of the non-lytic escape of Candida auris from macrophages that don’t rupture the phagosomal membrane.
Given the study findings, the scientists suggest that metabolic manipulation could function an efficient intervention to initiate phagocyte-mediated destruction of Candida auris and control infection.

 *Vital notice: bioRxiv publishes preliminary scientific reports that usually are not peer-reviewed and, due to this fact, mustn’t be thought to be conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Vital notice: bioRxiv publishes preliminary scientific reports that usually are not peer-reviewed and, due to this fact, mustn’t be thought to be conclusive, guide clinical practice/health-related behavior, or treated as established information.
Journal reference:
- Preliminary scientific report.
Candida auris evades innate immunity by utilizing metabolic strategies to flee and kill macrophages while avoiding antimicrobial inflammation, Harshini Weerasinghe, Claudia Simm, Tirta Djajawi, Irma Tedja, Tricia L Lo, David Shasha, Naama Mizrahi, Françios AB Olivier, Mary Speir, Kate E. Lawlor, Ronen Ben-Ami, Ana Traven, bioRxiv 2023.02.28.529319; doi: https://doi.org/10.1101/2023.02.28.529319, https://www.biorxiv.org/content/10.1101/2023.02.28.529319v1